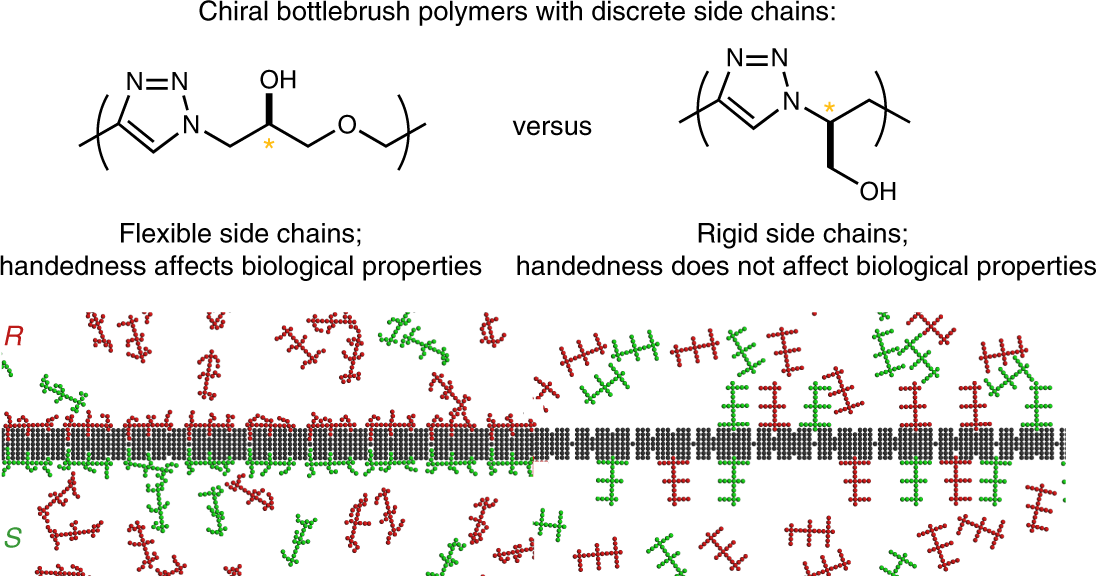
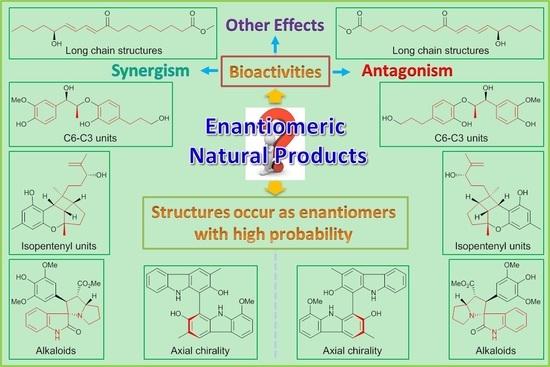
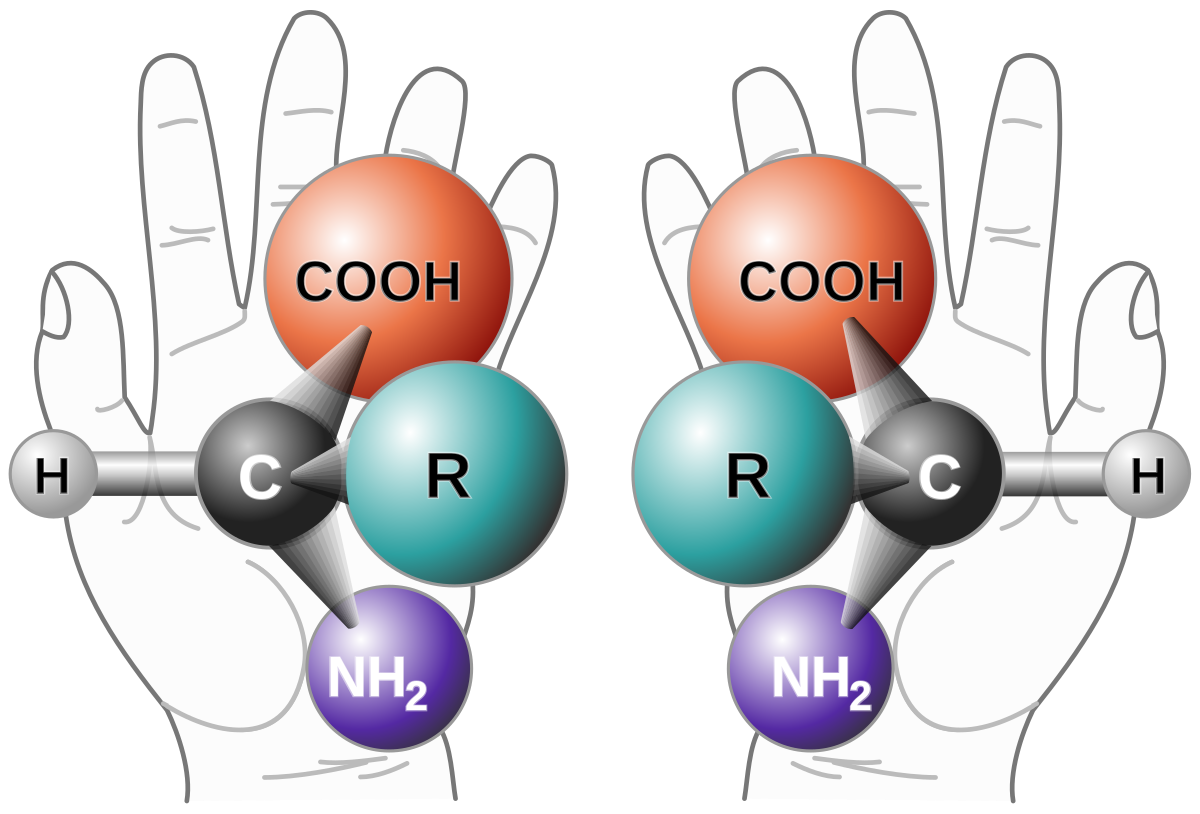
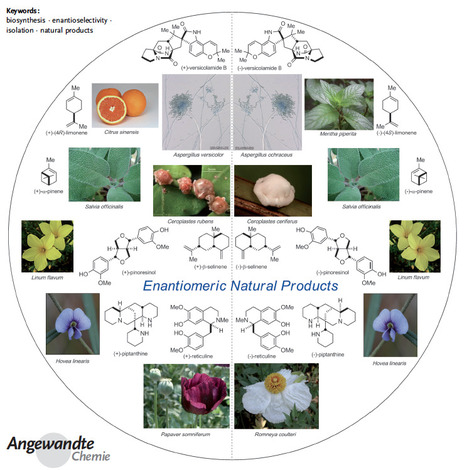
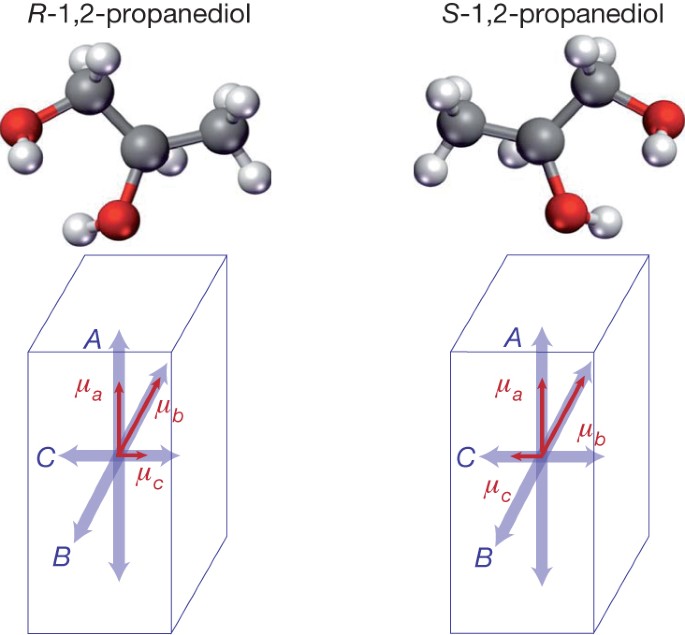
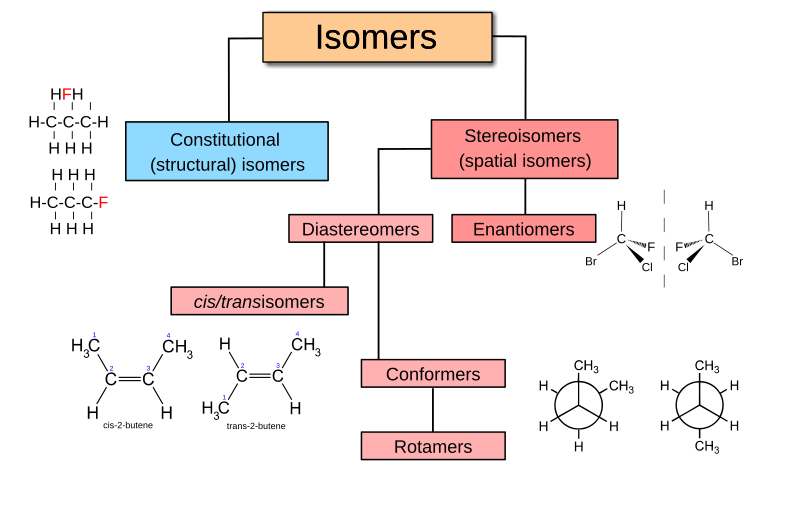
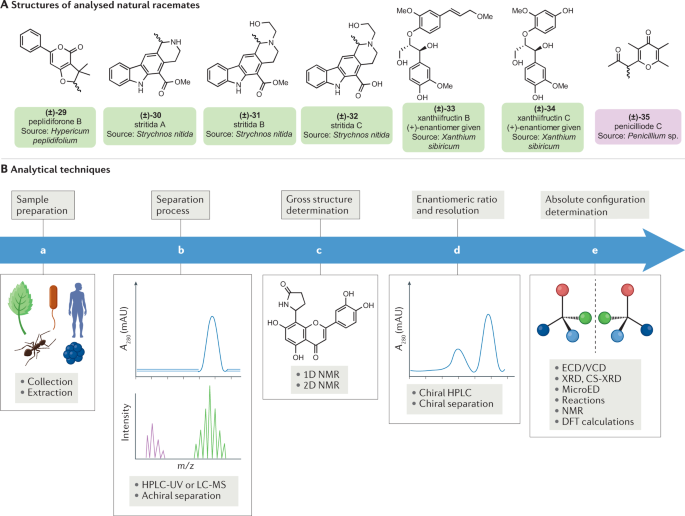
A tribute to Jacobus van 't Hoff: Enantiomers become diastereomers in optically active solvents. | Nature Portfolio Chemistry Community

Bilayer-dependent inhibition of mechanosensitive channels by neuroactive peptide enantiomers | Nature

Handedness-dependent quasiparticle interference in the two enantiomers of the topological chiral semimetal PdGa | Nature Communications

Enantiomer-selective magnetization of conglomerates for quantitative chiral separation | Nature Communications
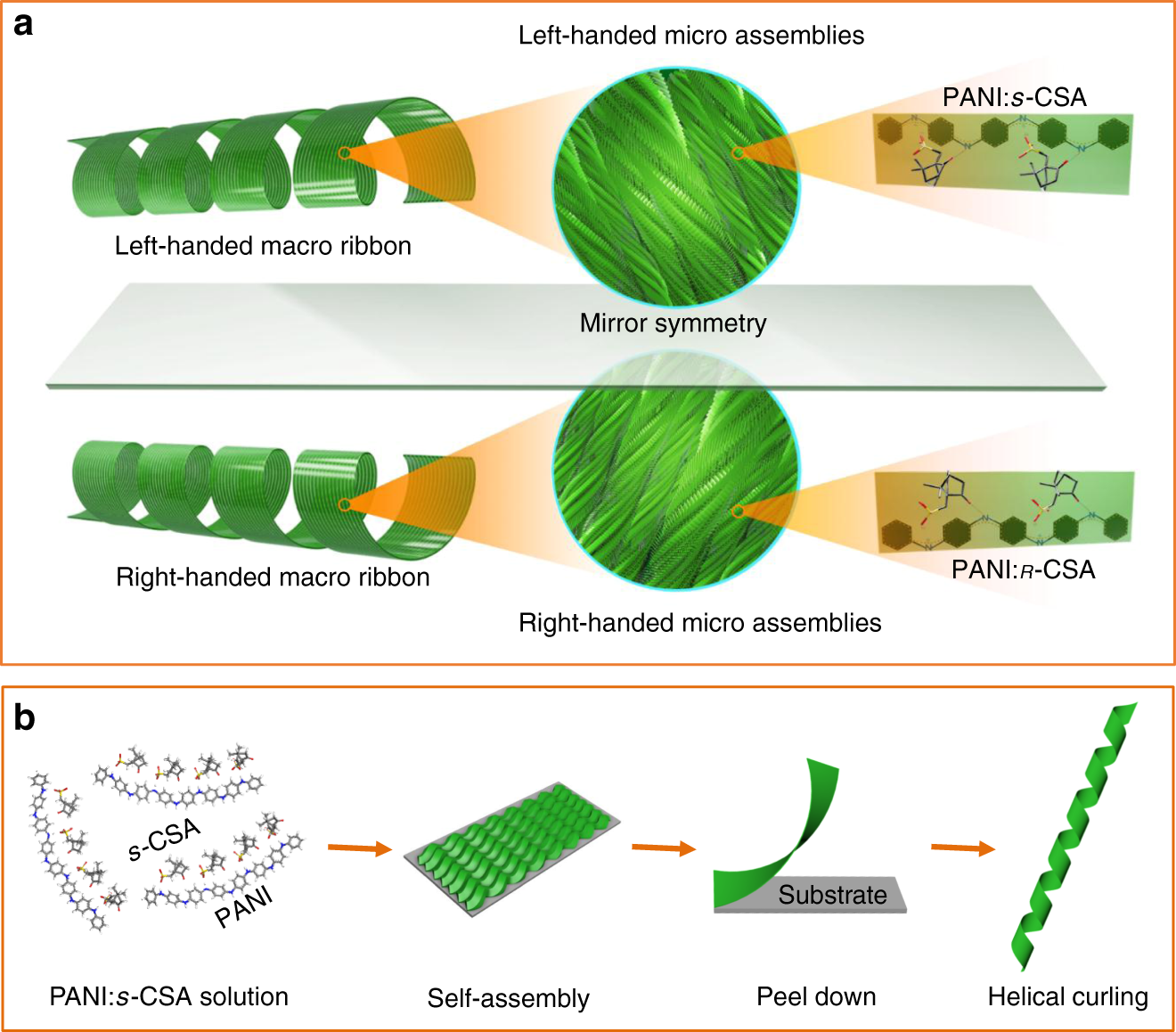
Macroscopic helical chirality and self-motion of hierarchical self-assemblies induced by enantiomeric small molecules | Nature Communications
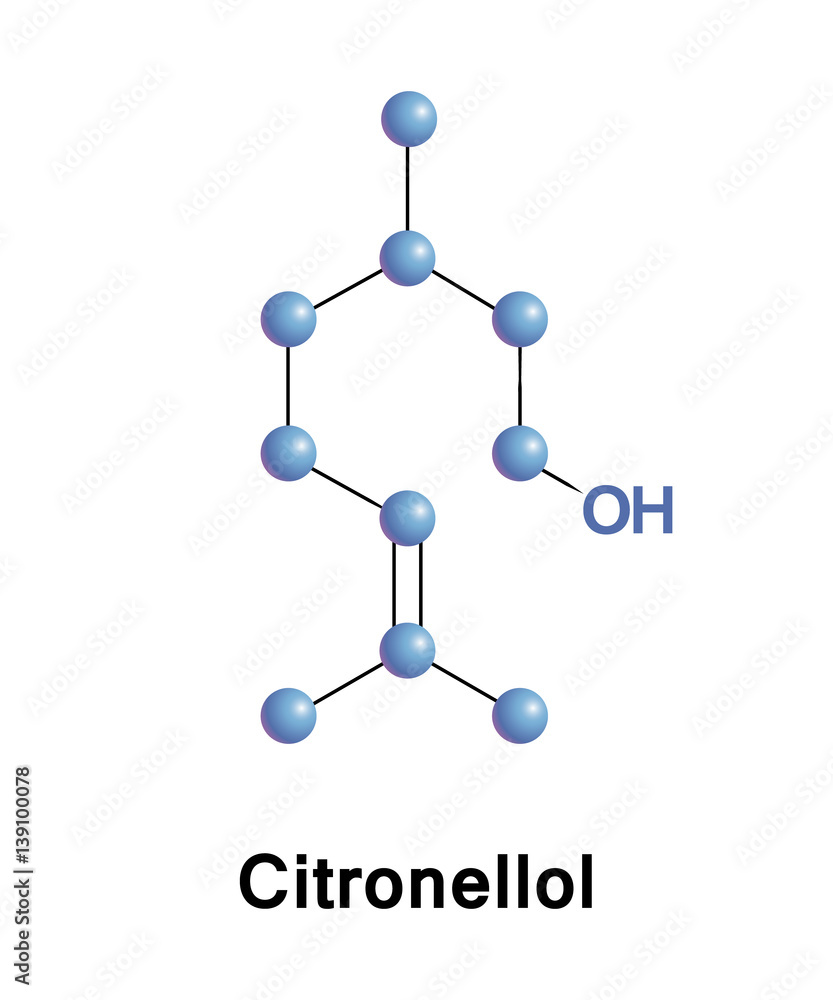
Citronellol, or dihydrogeraniol, is a natural acyclic monoterpenoid. Enantiomers occur in nature. It is found in citronella oils, including Cymbopogon nardus, is the more common isomer. Stock Vector | Adobe Stock